Helical Polymers: Catalysis
Inspired by experiments in the 90ies to use not only solid phase synthesis but also soluble polymers for combinatorial chemistry, the idea was born to develop soluble, helically chiral polymers as ligands for asymmetric transition metal catalysis. The multiple repetition of catalytically active sites in a stereoregular microenvironment should lead to an asymmetric multiple-site catalyst, combining process advantages related to its macromolecular state (easy separation, recyclability) with chemical function (turn-over and asymmetric induction). Especially attractive appeared the possibility to use the helicity of the polymer backbone as the sole source of asymmetric induction.
To realize the goals described above, we started working with polymethacrylates and polyisocyanates. Possible applications of polyarylacetylenes and polyarylisonitriles are currently under investigation.
Since the turn of the millennium, based on the work of Okamoto, we prepared uniformly configured, nitrogen containing polymers and copolymers by helix-sense-selective anionic polymerization using a combination of chiral bases (DPEDA-Li and (+)-PMP).
Polyisocyanates
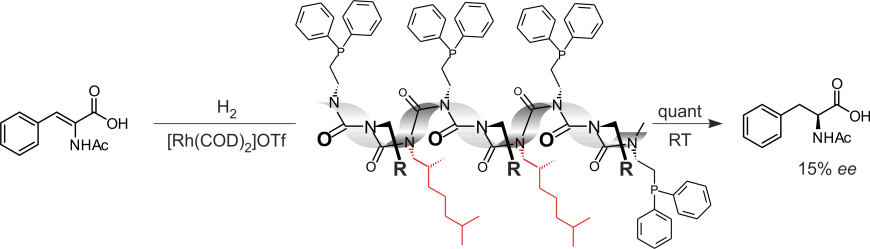
As representatives of dynamically helical polymers, polyisocyanates display a number of interesting properties (majority rules polymers; sergeant&soldiers polymers; dilutetd majority rules polymers: Green 1999). Based on these properties we synthesized sergeants&soldier polymers mit achiral phosphanylated soldiers. As chiral sergeant enantiomerically pure 2,6-dimethylheptylisocyanate was used. The optically active, helically chiral polymer was employed as a ligand for rhodium and the resulting complex was investigated as catalyst for asymmetric hydrogenations (PNAS 2004).
Current work in the field of helically chiral polymers as ligands for asymmetric transition metal catalysis focus on the application of polyacetylenes and polyisonitriles for this purpose.
At the same time these two latter polymer types will be developed further as chiral orienting media.